|
© Borgis - Postępy Nauk Medycznych 9, s. 714-724
Tomasz Dębski, Lubomir Lembas, *Józef Jethon
Basal cell carcinoma. Current views (Part III). Surgical treatment
Rak podstawnokomórkowy skóry. Współczesne poglądy (Część III). Leczenie chirurgiczne
Department of Plastic Surgery, The Medical Centre of Postgraduate Education in Warsaw
Head of the Department: Józef Jethon, MD, PhD, Professor of Plastic Surgery Streszczenie
Basal Cell Carcinoma (BCC) is one of the most common human malignancies. Although it is not directly life-threatening and rarely metastasises, its local malignant features cause significant functional and aesthetic disturbances, what has a profound effect on the quality of life of patients. The constantly increasing incidence of BCC observed within the last couple of years and the fact that this malignancy tends to occur in younger and younger people prove that BCC is now and will be in the future an important clinical problem. However, this problem is undernoticed and underestimated mainly to its low mortality and the fact that this malignancy is not listed in incidence reports. Although currently several factors are suspected to be responsible for BCC the most important roles in cancerogenesis are played by UV radiation and advanced age of patients. They account for more than 90% of BCC. Diagnosis of BCC basing only on the clinical appearance is difficult and depends on physician´s experience to a large extent. Traditionally, several clinical forms of BCC can be distinguished; however, the awareness of their existence may only help distinguish oncologically suspicious lesions, and not diagnose them. A histopathological examination is the only test that can verify and complete the BCC diagnosis. The clinical course of BCC is not characteristic and cannot be predicted: a lesion may not change for years, it may grow slowly or extremely fast, the infiltration area may enlarge or recede, it may also ulcerate or tend to heal. BCC metastases occur extremely rarely (<0.1% of cases). Słowa kluczowe: basal cell carcinoma, classification, epidemiology, pathogenesis, clinical appearance, metastases
Summary
Rak podstawnokomórkowy skóry (Basal Cell Carcinoma – BCC) jest najczęściej występującym nowotworem złośliwym człowieka. Chociaż nie zagraża on bezpośrednio życiu i rzadko daje przerzuty, jego miejscowa złośliwość jest przyczyną znacznych zaburzeń funkcjonalnych i estetycznych, co istotnie wpływa na jakość życia pacjentów. Stały wzrost zachorowań na BCC obserwowany w ciągu ostatnich lat oraz tendencja do występowania tego nowotworu u osób coraz młodszych, świadczą o tym, że BCC stanowi i będzie stanowił istotny problem kliniczny w przyszłości. Problem ten jest jednak niedostrzegany i niedoceniany głównie ze względu na niewielką śmiertelność oraz nieuwzględnianie tego nowotworu w zestawieniach zachorowalności. Chociaż w chwili obecnej postuluje się kilka czynników odpowiedzialnych za powstawanie BCC, najistotniejszą rolę w kancerogenezie odgrywają promieniowanie UV oraz zaawansowany wiek chorych. Odpowiadają one za ponad 90% zmian nowotworowych. Rozpoznanie BCC na podstawie samego obrazu klinicznego jest trudne i w dużej mierze zależy od doświadczenia badającego. Tradycyjnie wyróżnia się kilka form klinicznych BCC, których znajomość może pomóc jedynie w wyodrębnieniu zmian podejrzanych onkologicznie, a nie w rozpoznaniu. Jedynym badaniem weryfikującym rozpoznanie wstępne i decydującym o rozpoznaniu BCC jest badanie histopatologiczne. Przebieg kliniczny BCC jest niecharakterystyczny i nieprzewidywalny: zmiana może nie zmieniać się przez lata, rosnąć powoli lub bardzo szybko, zwiększać obszar nacieku lub ustępować, ulegać owrzodzeniu lub nawet goić się. Przerzuty BCC zdarzają się niezwykle rzadko (<0,1% przypadków). Key words: rak podstawnokomórkowy skóry, klasyfikacja, epidemiologia, patogeneza, obraz kliniczny, przerzuty
Surgical excision with a margin of clinically normal surrounding tissues
Surgical treatment is the simplest, the most effective and nowadays the most popular method of treatment. Its efficacy evaluated as the recurrence rate in a 5-year follow-up is 2-10% (1, 2, 3, 4, 5). So far only few prospective studies comparing surgical excision with other methods have been published in the literature.
Thissen compared cryosurgery (spray technique and double cycle of freezing) with surgical treatment of nodular and superficial BCC of the head and neck with the diameter of <2 cm and observed better therapeutic outcomes in surgically treated patients; however, the differences were not statistically significan (6).
On the other hand, Rhodes compared surgical treatment of nodular BCC located on the face with photodynamic therapy (PDT). He did not observe significant differences in recurrence rates during the first 3 months; however, in 12- and 24-month follow-up he observed a statistically significant increase in the recurrence rate and worse cosmetic results in the case of PDT (7). Taking into account long-term results (after 60 months), recurrence occurred in 14% after PDT and only in 4% after surgical treatment (8).
Moreover, another study where surgical excision of primary BCCs with the diameter below 4 cm was compared with radiation therapy indicated that during a 4-year follow-up a lower recurrence rate was associated with surgical excision (0.7% vs 7.5%.) Moreover, cosmetic results after surgical excision were more acceptable than the ones after radiation therapy (79% vs 40%), which were associated with discolouration and telangiectasia in more than 65% and radiodystrophy in 41% of cases (9, 10).
An important stage in surgical excision of a lesion is the determination of a macroscopic (clinical) lesion border. It can be done using magnification (3x at least), Wood´s lamp or dermatoscope (11). Applying curettage before excision may increase the completeness of a procedure because it may be possible to determine real borders of a tumour more precisely (neoplastic tissues are more curettable) (12, 13). When lesion borders have been precisely determined a margin (range) of clinically normal tissue is planned, and they are removed together with a lesion en-block. The specimen resected in this way is subject to histopathological evaluationduring which a histopathological subtype of BCC is confirmed as well as procedure completeness. The tissue defect formed after lesion excision is closed according to the reconstructive ladder (diagram 1) (fig. 1).
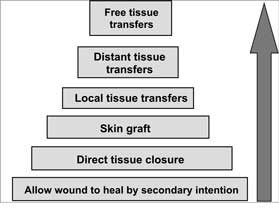 Diagram 1. The reconstructive ladder (14).
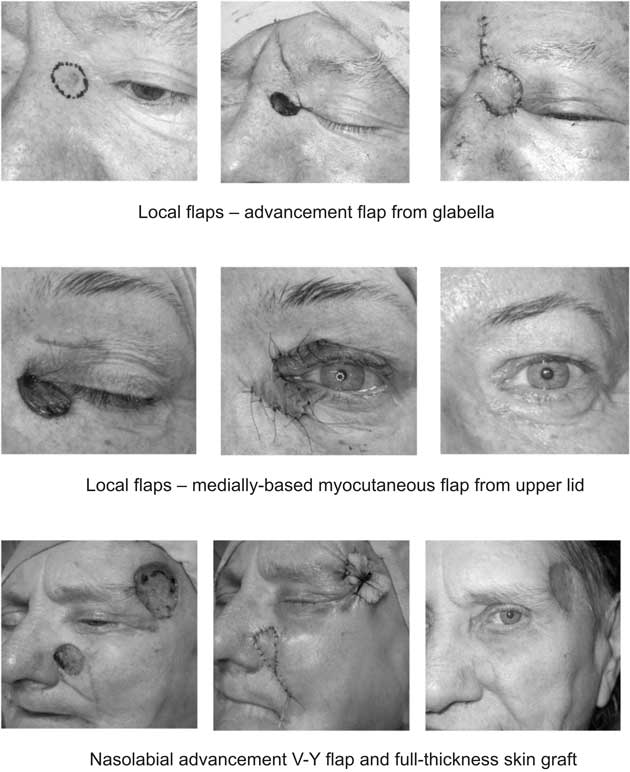 Fig. 1. Reconstruction methods in surgical treatment of BCC – examples.
Margin of clinically normal surrounding tissue
It is obvious that the extent of neoplastic infiltration affects the range of a peripheral and deep margin. On the other hand, the infiltration extent correlates with prognostic factors (Part I) (15) e.g. for primary morpheaform BCC resected with a 3-mm margin only 66% of radical excisions observed, with a 5-mm margin – 82% and with a 13-15-mm more than 95%. For that reason, the presence of prognostic factors should determine the extent of a margin. Nonetheless, a precise therapeutic algorithm has so far not been established (15).
The extent of suggested margins ranges from 2 to 15 mm (tab. 1), is established empirically and what is the most important, it does not take prognostic factors into account.
Table 1. Range of excision margin – analysis of selected studies.
As reports published so far are in the majority retrospective reviews of therapeutic outcomes for different margins, their conclusions should be treated more like advices rather than methodologically proven guidelines.
Although American recommendations of NCCN (National Comprehensive Cancer Network) (Part II) recommend to remove low-risk BCCs with a 4-mm margin it has to be noted that, what is emphasised by the authors of these recommendations, this guideline was based on lower-level evidence (16). It is based on the prospective report by Wolf from 1987 (tab. 1) where 117 primary lesions were resected with a 2-mm margin with subsequent histopathological evaluation according to Mohs. If excision was incomplete, the margin of resected tissues was expanded by 1 mm until the procedure was complete. When lesions were excised with 2-mm margins, completeness of 70% was achieved, with 3-mm margins excisions were complete in 85% of cases and when margins were 4 mm completeness was as high as 95%. Although these results are statistically significant, they are poorly reliable because the study did not take into account prognostic factors that have been identified until now. The study lacks in information regarding the site of a tumour, its histopathological subtype and regards only well-demarcated lesions, and in 91% of cases the tumour diameter was below 2 cm, and the mean was 9 mm. Due to low reliability of studies published so far the need to perform prospective studies according to evidence based medicine (EBM) arises as it is necessary to determine unanimously the range of margins depending on prognostic factors.
It is of special importance when BCCs are located in functionally and aesthetically important face areas such as nose (ala nasi, columella, naso-facial sulcus), eyelids, medial and lateral canthus, ear with preauricular and postauricular region, lips philtrum (17, 18). All anatomical regions mentioned above are associated with a high risk of recurrence (H-area – Part II), and the differences in suggested margin are extremely important although they are as small as several millimetres.
The excision of a lesion with a larger margin may for example result in the removal of lacrimal ducts or cause ectropion. For that reason, during margin selection a surgeon resecting a lesion in these areas (a suggested margin for the face is from 2 to 10 mm) has to take into account such aspects as post-surgical function preservation, possibility of defect closure or, last but not least, achieving acceptable cosmetic results.
As it can be seen, due to varied and often not very reliable recommendations, the decision to choose an oncologically radical margin is extremely difficult and in most cases made based on operator´s experience (his knowledge and skills).
Prospective trials taking into account all factors affecting the range of a margin (including prognostic factors) would systematise the current state of knowledge and contribute to establishing consensus regarding the excision margins, what is indispensable in order to prepare a consistent algorithm for BCC treatment.
The deep margin range is less controversial. The majority of authors recommend to resect BCCs at the depth reaching the fat tissue (15). In BCCs where the skin is directly above deep tissues (cartilages, bones) it is obligatory to perform imaging tests and if necessary, increase the depth of a margin (19).
Histopathological evaluation
In order to determine the location of a resected tumour with relation to the adjacent tissues, each specimen indicated for a histopathological examination should be equipped with a surgical marker (suture, cut, dye mark) and a precise diagram presenting its location with relation to the adjacent tissues. As a result, it will be possible to identify sites where excision was incomplete. Moreover, information regarding previous treatment and results of a biopsy are also necessary (19).
The aim of a histopathological examination is to determine a histological subtype of BCC and excision completeness.
From a histopathological point of view BCC is similar to the basement membrane cells but differs in that the nucleus/cytoplasm ratio is higher.
Moreover, BCC cells do not have intercellular bridges and mitotic figures (20).
According to the latest WHO classification there are several clinical-pathological types of BCC. It includes criteria of a clinical and histopathological picture of different subtypes of BCC (21).
From a clinical point of view it is important to distinguish histopathological subtypes with a aggressive growth pattern, what is of importance when a surgical procedure is planned (tab. 2). In addition, infiltration along vessels and nerves is dangerous, and associated with a high risk of recurrence (11).
Table 2. Clinical-pathological classification of BCC (WHO 2006) and classification based on the recurrence risk (21, 22).
The literature also describes undifferentiated and differentiated BCC. Little or no differentiation is reffered to as a solid BCC and includes pigmented, superficial, morpheaform and infiltrative subtypes. Differentiated BCC is mainly nodular BCC which often differentiates into cutaneous appendages, including hair, sebaceous or tubular glands (20). For statistical purposes the classification including three types is often used: nodular (solid), fibrosing (desmoplastic) and metatypic (baso-spinocellular) (22).
Based on precise histopathological evaluation it was determined that neoplastic infiltration is irregular and unpredictable; however, it is usually present to a limited extent (23).
The margin of clinically normal surrounding tissues viewed under a microscope is on average smaller by 24% from a macroscopic in vivo margin as a result of shrinking and fixing (24).
Hendrix et al. proved that infiltration in the case of infiltrating BCC is on average 7.2 mm, whereas for nodular BCC it is 4.7 mm (primary /recurrent 5.6/10.4 vs 4.3/5.7 respectively) (25). On the other hand, according to Salasche primary morpheaform BCC can infiltrate tissues even up to the width of 7 mm beyond macroscopic borders of a lesion (26). Based on Burg´s studies microscopic borders of primary BCC infiltration are estimated to be 3-6 mm, and 5-9 mm in the case of recurrent disease (23).
The most important factor determining the recurrence is to determine the extent of neoplastic infiltration and its relation to the edge of resection (19). It regards cases where tumor was identified at peripheral edge of resection and deep edge of resection what is associated with a more than a double risk of recurrence (17% peripheral vs 35% deep surgical edge in a 5-year follow-up period) (27).
In order to evaluate the risk of recurrence Pascal (28) made the following classification:
? incomplete excision – a tumor is identified at a surgical edge. It is associated with a 33% risk of recurrence within 5 years follow-up.
? suboptimal excision – tumor is visible at the distance smaller than 0.5 mm from a surgical edge, what microscopically corresponds to one high-power field (400x). In such cases there is a 12% risk of recurrence within 5 years follow-up.
? complete excision – tumor is visible at the distance larger than 0.5 mm, namely it is not present in one high-power field (400x) involving a surgical edge.
Although resecting a tumor in these cases is described as complete excision it is associated with a 1.2% risk of recurrence within 5 years follow-up.
It is mainly associated with the fact that histopathological techniques are not perfect. Conventional microscoping processing involves vertical sectioning at every 2-4 ?m, perpendicular to the tumor surface (breadloaf sectioning -recommended for lesions <16 mm) (29), when specimens from all four lesion quadrants are resected (cross sectioning – recommended>16 mm) (29) or from its peripheral parts (peripheral sectioning) (17) (fig. 2A, B, C). Consequently, only representative vertical sections are evaluated and it is not possible to evaluate the whole tumor margin. It is claimed that using conventional methods it is possible to evaluate only from 0.01% to 44% of tumor margins (17, 30).
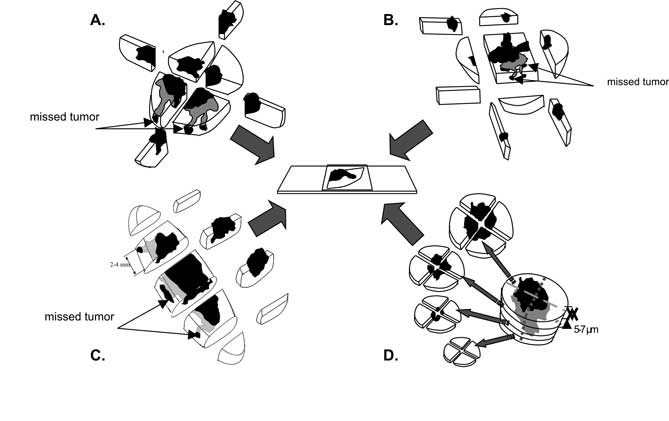 Fig. 2. Methods of microscopic processing: cross sectioning (A), peripheral sectioning (B), breadloaf sectioning (C), Mohs method (D).
The basic drawback of this method is an assumption that when a tissue margin in representative sections is considered as tumor-free, the whole lesion is considered to be excised completely. The method suggested by Mohs is significantly more precise than conventional techniques as it allows for collecting sections which are horizontal to the tumor surface, namely it is possible to evaluate the whole margin (fig. 2D).
Incomplete excision
Incomplete excision of BCC is a situation when a tumor is identified at surgical edge or is at the distance of <0.5 mm from it (one high-power field at 400x magnification). On average 4.7%-7% (31, 32) of cases in the UK and 6.3% in Australia (33, 34) are excised incompletely. These numbers prove that a problem is huge and although factors responsible for an increased risk of incomplete excision have already been identified (Part II) until now it has not been possible to create a consistent algorithm of BCC management.
The necessity to improve procedure completeness is undeniable; however, management following incomplete excision (follow-up, reexcision, radiation therapy) raises many controversies.
Followers of follow-up base their opinions on numerous studies proving that recurrent disease will occur only in 30-41% of cases (34, 35, 36) out of all incompletely excised lesions, so as many as 2/3 of tumor tissue left in the skin will not recur.
In addition, prospective studies where incompletely
excised BCCs were reexcised showed the presence of tumor tissue only in 45% (37) and 54% (32) using conventional histopathological evaluation and 55% using the Mohs method (38). It means that in almost half of cases the tumor tissue which was left regresses, possibly with the help of the immune system (38).
Is a reexcision of an incompletely excised tumor justified?
Richmond et al. included in their study 92% patients who underwent reexcision of an incompletely excised tumors and 90% patients who were only observed (until recurrence occur, then it was excised immediately). After a 10-year follow-up period recurrence occurred only in 9% of patients in the first group and in as many as 60% of patients in the other (39).
Koplin recommends reexcision only in two cases: when a tumor is identified at surgical edge and when a tumor was excised suboptimally; however, only when expected life-time is long (40).
On the other hand, Robinson recommends only follow-up in the case of incomplete excision of an aggressive histopathological subtypes of BCC located on the nose, cheeks, around the lips, in men>65 years and in the case when a flap or split-thickness skin graft was used to cover a defect. His stand is based on the observation that in cases mentioned above significantly longer time is required for recurrence development (>5 years) than in other cases of BCC excised incompletely (41).
Similar controversies are associated with the use of radiation therapy following incomplete BCC excision. In a 10-year follow-up Liu compared the number of recurrence in the group of patients after immediate post-operative radiation therapy with the group of observed patients. Although in the group of patients subject to post-operative radiation therapy the recurrence rate was significantly lower the evaluation of all treatment aspects (costs analysis, complications) indicated that it is only recommended in cases after recurrence and when deep and lateral margins are involved. In the remaining cases the authors recommend careful observation stating a high presence of patients on follow-up examinations and significantly lower treatment costs as arguments for it (27).
On the other hand Wilson et al. recommend radiation therapy or reexcision of a tumor following its incomplete excision when a local flap or full-thickness skin graft was applied and when a deep margin in the H zone (Part III) was involved. In all such cases recurrence would be difficult to detect and treat. In addition, Wilson does not recommend radiation therapy in young patients, especially in the forehead and scalp due to the risk of neoplastic transformation in scarred lesions in a long-term follow-up. In such cases, as well as in the cases of histologically aggressive BCC reexcision is recommended (37).
Some authors have even more restrictive views on radiation therapy and they recommend it only in small lesions in patients who do not agree surgical treatment or in whom it is not possible to perform a surgery due to their physical condition (19).
Recurrence
Recurrence is defined as a new focus of tumor in a scar or in its vicinity formed after excision of a primary tumor occurring within 5 years since the excision and with the same histological type as the primary tumor. The incidence rate after surgical treatment of BCC in a 5-year follow-up is estimated to be 5% for primary and about 13% for recurrence BCC (42).
The factors increasing the risk of recurrence have been discussed in the Part II. Recurrence is the most frequent, namely as many as 82% of cases occur in the first five years, and about 30% of cases are present in the first year of observation, 50% in the second and 66% in the third year. The remaining 18% of cases occur in the period from 5 to 10 years since tumor excision (44). The recommended follow-up for one patient in order to observe recurrence is 5 years (every 3 months during the first year, then every 6 months) (27) although the latest guidelines of the American NCCN recommend follow-up every 6-12 months for the whole lifetime (16). During follow-up control it is necessary to educate a patient with the possibilities of active prophylaxis against BCC, especially regarding sun protection. Moreover, it is also necessary to inform a patient about the need of skin self-examination. It is of special importance not only because of recurrence monitoring but also due to the risk of development of another BCC which is ten times higher in patients with a previous BCC than in the general population (44). This risk is also significantly higher in elderly patients, in patients with multiple BCCs and with the lesion diameter> 1 cm (45).
In some countries general practitioners manage follow-up control after BCC excision (46).
Clinical symptoms that should raise suspicion of recurrence:
? scarring with non-healing or recurrent ulceration,
? a scar that is becoming red, desquamating and looks like ichtyosis,
? an expanding scar with telangiectasia inside the scar,
? a nodule appearing within a scar,
? tissue destruction.
Recurrence following non-surgical treatment (radiation therapy, cryotherapy, curettage) is associated with a higher risk of another recurrence or even with the possibility of metastases (47).
Skin lesions due to such treatment (atrophy, hypopigmentation, scarring) make it difficult to assess the extent of recurrence precisely. In addition, recurrence following treating BCC with cryotherapy may be difficult to evaluate due to scarring associated with treatment and the fact that infiltration spreads deeply beneath normally looking skin (19). Moreover, recurrence diagnostics in skin creases such as the nasolabial sulcus may also be a huge problem. Due to diagnostic difficulties in all cases when recurrence is suspected it is necessary to perform a biopsy to confirm diagnosis.
Recurrence treatment is significantly more difficult than treatment of a primary lesion, and the success rate is considerably lower (48). The risk of another recurrence is by 50% higher that in the case of a primary lesion (5). Furthermore, it is thought that some incompletely excised lesions recur in a more aggressive growth pattern (a more aggressive histological subtype), especially in the central face (49).
Surgical methods are recommended to treat recurrence. Wide BCC excision with a scar and a margin ranging from 5 to 10 mm or Mohs micrographic surgery which is significantly more successful are performed.
Mohs micrographic surgery (excision controlled histopathologically) (17, 51)
The history how this method came into existence and evolved is tightly associated with its creator. In 1930s Frederic E. Mohs, who was a medicine student at those times, studied the effects of different substances on cancer cells in rats. When he injected 20% zinc chloride solution inside a tumour he accidentally observed tissue necrosis and at the same time in situ tissue fixation. As a result of in vivo tissue fixation with zinc chloride he was able to cut specimens horizontally (parallel to the skin). As a result he was able not only to assess a tumor histopathologically, but also to assess its whole margin, what formed the base to excision of tumors under microscopic control. After a short period of laboratory tests Mohs replaced 20% zinc chloride solution with the zinc paste applied 24h before tumor excision and started clinical trials on the use of his method in the treatment of skin tumors. As early as in 1941 Mohs presented his first clinical trials in „Archives of surgery” and named his method chemosurgery (50). (chemo – as tissue fixation is a result of a chemical reaction between tissues and zinc chloride) (diagram 2).
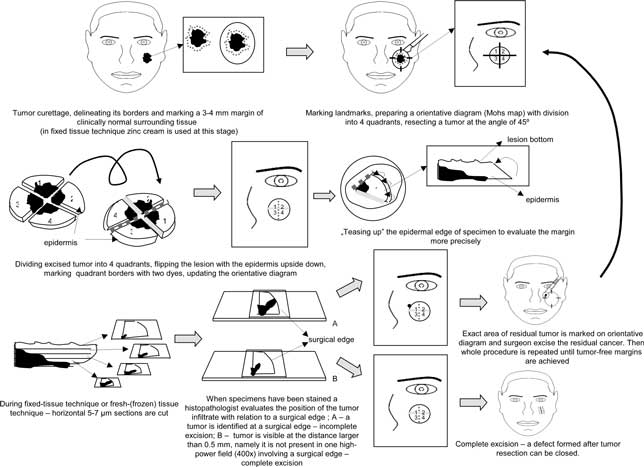 Diagram 2. Mohs micrographic surgery.
It involves curettage of a tumour then applying a thick layer of the zinc paste under occlusive dressing which increases paste penetration and protects the surrounding skin. In the case of tumors with large hyperkeratosis before applying the zinc paste dichloroacetic acid with the keratolytic effects can be used and as a result, after the keratotic layer has been exfoliated, penetration of the zinc paste is greater. Tissues prepared in this way become necrotic after 6-24 hours what creates appropriate conditions for a surgical procedure (no pain, small bleeding at the operation site). During the first stage a surgeon uses methylene blue, sutures or incisions to mark landmarks that will allow further identification of tumor areas that have been excised incompletely. Then the whole tumor with a 3-mm margin of clinically normal surrounding tissues is excised at the angle of 45°, what makes it possible to „tease up” a epidermal edge and facilitates histopathological evaluation, especially of superficial skin layers. The incision is continued at the angle of 45° around the tumor, and the deep surgical edge is parallel to the skin. After haemostasis a diagram presenting a defect formed after tumor excision and its relation to previously marked landmarks is prepared.
In the next stage, a histopathologist divides a lesion according to landmarks marked by an operator (usually into 4 quadrants), turns it deep surface up and marks edges of all quadrants formed after the division with two different dyes. Consequently, further identification of specimen areas excised incompletely is made easier. After marking quadrant edges the specimen is sectioned parallel to the skin and as a result 5-7 μm sections are obtained, therefore it is possible to assess the whole lesion margin. When a tumor fragment is found in the close vicinity of a surgical edge (suboptimal excision) or when a tumor is identified at surgical edge (incomplete excision) a histopathologist marks an area of incomplete excision in the diagram. Based on this diagram a surgeon locates the area with tumor cells that has been left, and then the whole process of fixation, excision and histopathological evaluation is repeated until complete excision is obtained.
In some cases when lesions were excised incompletely the whole process was repeated several times, what due to long time of tissue fixation with the zinc paste (6-24h) took even several days (high costs of treatment and hospitalisation). Moreover, application of the zinc paste was painful for patients and induced tissue necrosis and ulceration in the lesion vicinity.
Therefore after tumor excision it was necessary to wait until necrosis areas became defined, and only then it was possible to close formed defect. All these features resulted in the fact that despite high efficacy this method did not gain much popularity. However, Mohs, who believed in the success of his method, focused more on its promotion than improvement. And in 1953 when he was doing a film demonstrating his technique an accident decided about a breakthrough discovery for yet another time. Mohs during resecting BCC of the lower eyelid omitted the stage of tissue fixation with the zinc paste in order to accelerate the whole process for the purposes of the film. Instead he excised BCC under local anaesthesia, then froze it and using cryotome obtained horizontal specimens of 5-7 μm. After their standard staining with haematoxylin-eosin he stated than the quality of specimens obtained in this way is the same when compared to the original method, and the duration of the procedure, costs and lack of ulceration and pain are significantly lower. Moreover, primary defect closure was possible what decided about the success of this method to a large extent. With time the fresh-tissue technique replaced the fixed-tissue technique that was originally developed by Mohs. The original name of the whole procedure (chemosurgery) was replaced by Mohs Micrographic Surgery (MMS) due to unfavourable associations and in order to confer its meaning, namely mapping out of the specific location of tumor extension by the use of microscopic evaluation (diagram 2).
Currently the fixed-tissue technique is used only in selected cases, mainly in order to eliminate excessive bleeding during lesion excision in areas which are well supplied with blood (e.g. penis.) The fresh-tissue technique has considerably more indications in BCC treatment (tab. 3).
Table 3. Indications for Mohs Micrographic Surgery (at least one of the mentioned above) (17).
A review of studies published since the mid-1940s confirms the high efficacy of this method reaching even 99% in a 5-year follow-up for primary (52) and 94.4% for recurrent BCC (48).
In prospective trials conducted in Australia in 819 patients with periocular BCC no recurrence were observed in 100% patients with primary and in 92.2% with recurrent BCC (53) in a 5-year follow-up, and in another study with 3370 patients with BCC on the head and neck no recurrence were observed in 98.8% patients with primary BCC and in 96% with recurrent BCC in a 5-year follow-up (54).
Apart from its high efficacy other advantages of this method include the minimum amount of excised tissues and the possibility of primary defect closure knowing that a lesion was excised completely. Despite many advantages Mohs Micrographic Surgery has been fully approved only in the USA, where it is used in about 30% of cases (55).
Also in the USA was formed the American College of Mohs Micrographic Surgery and Cutaneus Oncology that currently has more than 800 physicians of different specialities who are qualified to use this method when they have completed a 1-2 year training course. In other countries Mohs micrographic surgery is used more rarely, mainly due to high costs (equipment, service, qualified staff) (56) and more work contribution (57) (time of one procedure is 3h on average) compared to traditional surgical lesion excision. Moreover, contrary to a classic surgical procedure this method is laborious, time-consuming and exhausting for a patient.
Some researchers defend high costs of treatment using this method by the fact that it is possible to obtain better results, consequently, it is more effective in terms of treatment costs than classic surgery (57). However, Smeets in a prospective trial comparing this method with surgical excision of a lesion with a 3-mm margin of clinically normal surrounding tissue following 30 months of follow-up did not find statistically significant differences in the treatment efficacy between these two method (58).
BCC is one of the most common human malignancies. Epidemiology and the increasing rate of incidence prove that BCC is and will be in the future a significant clinical problem. Studies published recently complete our knowledge on its pathogenesis and present new diagnostic and therapeutic methods. The majority of new therapeutic methods may be used only in highly selected cases.
The only universal method that can be used to treat all cases of BCC is surgical excision with a margin of clinically normal surrounding tissues. However, despite numerous advantages of this method and its versatility it has been impossible to determine until now what margin of clinically normal surrounding tissue should be applied when BCC is excised. It is of special importance when lesions in functionally and aesthetically important face areas are excised. In such cases margin differences as low as 1 mm may affect completeness of excision, its functional results, the method of reconstruction and its aesthetic results as well.
For that reason, the authors think that prospective studies in accordance with EBM are necessary in order to determine unanimously the range of margins depending on the prognostic factors mentioned above.
The results of such studies could allow for the unanimous determination of the excision margins and preparing a consistent and common algorithm of BCC treatment.
Piśmiennictwo
1. Walker P, Hill D: Surgical treatment of basal cell carcinomas using standard postoperative histological assessment. Australas J Dermatol 2006; 47: 1-12.
2. Griffiths RW, Suvarna SK, Stone J: Do basal cell carcinomas recur after complete conventional surgical excision? Br J Plast Surg 2005; 58: 795-805.
3. Goldberg LH, Hsu SH, Alcalay J: Effectiveness of isotretinoin in preventing the appearance of basal cell carcinomas in basal cell nevus syndrome. J Am Acad Dermatol 1989; 21: 144-145.
4. Cullen FJ, Kennedy DA, Hoehn JE: Management of basal cell carcinoma: current concepts. Adv Plast Surg 1993; 10: 187.
5. Silverman MK et al.: Recurrence rates of treated basal cell carcinomas. Part 3: Surgical excision. J Dermatol Surg Oncol 1992; 18: 471-476.
6. Thissen MRTM, Nieman FHM, Ideler AHLB: Cosmetic results of cryosurgery versus surgical excision for primary uncomplicated basal cell carcinomas of the head and neck. Dermatological Surgery 2000; 26: 759-64.
7. Rhodes LE et al.: Photodynamic therapy using topical methyl aminolevulinate vs surgery for nodular basal cell carcinoma. Arch Dermatol 2004; 140: 17-13.
8. Rhodes LE et al.: Five year follow-up of a randomized, prospective trial of methyl aminolevulinate photodynamic therapy vs surgery for nodular basal cell carcinoma. Arch Dermatol 2007; 143: 1131-6.
9. Avril MF et al.: Basal cell carcinoma of the face: surgery or radiotherapy? Results of a randomized study. Br J Cancer 1997; 76: 100-6.
10. Petit JY et al.: Evaluation of cosmetic results of a randomized trial comparing surgery and radiotherapy in the treatment of basal cell carcinoma of the face. Plast Reconstr Surg 2000; 105: 2544-51.
11. Costantino D, Lowe L, Brown DL: Basosquamous carcinoma – an under-recognized, high-risk cutaneous neoplasm: case study and review of the literature. J Plast Reconstr Aesthet Surg 2006; 59: 424-8.
12. Johnson TM, Tromovitch TA, Swanson NA: Combined curettage and excision: a treatment method for primary basal cell carcinoma. J Am Acad Dermatol 1991; 24: 613-17.
13. Chiller K et al.: Efficacy of curettage before excision in clearing surgical margins of nonmelanoma skin cancer. Arch Dermatol 2000; 136: 1327-32.
14. Aston SJ, Beasley RW, Thorne CH: Grabb and Smith´s Plastic Surgery, 5th Edition, Philadelphia, Lippincott-Raven 1997.
15. Telfer NR, Colver GB, Morton CA: Guidelines for the Management of Basal Cell Carcinoma. Br J Dermatol. 2008; 158(7): 35-48.
16. Clinical Practice Guidelines in Oncology: Basal Cell and Squamous Cell Skin Cancers V.I.2009: www.nccn.org
17. Mohs Mooney M, Parry E. Mohs Micrographic Surgery 2007: www.emedicine.com/derm/topic542.htm
18. Chicheł A, Skowronek J: Współczesne leczenie raka skóry – dermatologia, chirurgia czy radioterapia? Współczesna Onkologia 2005; 9, 10: 429-435.
19. Australian Cancer Network Management of Non-Melanoma Skin Cancer Working Party. Non-melanoma skin cancer: Guidelines for treatment and management in Australia; 24 October 2002.
20. Bader RS, Basal Cell Carcinoma: Differential Diagnoses & Workup (2008): http://emedicine.medscape.com/article/276624-diagnosis
21. Kossard S et al.: Basal cell carcinoma. [In:] LeBoit P, Burg G, Weedon D, Sarasin A. The WHO classification of tumours. Pathology and genetics of skin tumours. IARC Press, Lyon 2006: 13-19.
22. Bieniek A, Cisło M, Jankowska-Konsur A: Nowotwory skóry. Klinika, patologia, leczenie, Galaktyka 2008.
23. Burg G et al.: Histographic surgery: accuracy of visual assessment of the margins of basal-cell epithelioma. J Dermatol Surg 1975; 1: 21-24.
24. Thomas DJ, King AR, Peat BG: Excision margins for nonmelanotic skin cancer. Plast Reconstr Surg 2003; 112: 57-63.
25. Hendrix JD, Parlette HL: Duplicitous growth of infiltrative basal cell carcinoma. Analysis of clinically undetected tumor extent in a paired case-control study. Dermatol Surg 1996; 22: 535-539.
26. Salasche SJ, Amonette RA: Morpheaform basal-cell epitheliomas. A study of subclinical extensions in a series of 51 cases. J Dermatol Surg Oncol 1981; 7: 387-394.
27. Liu FF et al.: A management approach to incompletely excised asal cell carcinomas of skin. Int J Radiat Oncol Biol Phys 199; 20: 423-428.
28. Pascal RR, Hobby LW, Lattes R: Crikelair GF, Prognosis of „incompletely” versus „completely excised” basal cell carcinoma. Plast Rec Surg 1968; 41; 328-332.
29. Rapini RP: Comparison of methods for checking surgical margins. J Am Acad Dermatol 1990; 23: 288-94.
30. Kimyai-Asadi A, Goldberg LH, Jih MH: Accuracy of serial transverse cross-sections in detecting residual basal cell carcinoma at the surgical margins of an elliptical excision specimen. J Am Acad Dermatol 2005; 53: 469-74.
31. Kumar P et al.: Jncidence of incomplete excision in surgically treated basal cell carcinoma: a retrospective clinical audit. Br J Plast Surg 2000; 35: 563-6.
32. Griffiths RW: Audit of histologically incompletely excised basal cell carcinomas: recommendations for management by re-excision. Br J Plast Surg 1999; 52: 24-8.
33. Dieu T, Macleod AM: Incomplete excision of basal cell carcinomas: a retrospective audit. Aust NZ J Surg 2002; 72: 219-21.
34. Sussman LA, Liggins DF: Incompletely excised basal cell carcinoma: a management dilemma? Aust NZ J Surg 1996; 66: 276-8.
35. Park AJ, Strick M, Watson JD: Basal cell carcinomas: do they need to be followed up? J R Coll Surg Edinb 1994; 39: 109-11.
36. De Silva SP, Dellon AL: Recurrence rate of positive margin basal cell carcinoma: results of a five-year prospective study. J Surg Oncol 1985; 28: 72-4.
37. Wilson AW et al.: Surgical management of incompletely excised basal cell carcinomas of the head and neck. Br J Oral Maxillofac Surg 2004; 42: 311-14.
38. Bieley HC, Kirsner RS, Reyes BA, Garland LD. The use of Mohs micrographic surgery for determination of residual tumor in incompletely excised basal cell carcinoma. J Am Acad Dermatol 1992; 26: 754-6.
39. Richmond JD, Davie RM: The significance of incomplete excision in patients with basal cell carcinoma. Br J Plast Surg 1987; 40: 63-67.
40. Koplin L, Zarem HA: Recurrent basal cell carcinoma: a review concerning the incidence, behavior, and management of recurrent basal cell carcinoma, with emphasis on the incompletely excised lesion. Plast Reconstr Surg 1980; 65: 656-64.
41. Robinson JK, Fisher SG: Recurrent basal cell carcinoma: A management dilemma? Aust NZ J Surg 1996; 66, 276-278.
42. Hauben DJ et al.: The biologic behaviour of basal cell carcinoma: analysis of recurrence in excised basal cell carcinoma. Part II. Plast Reconstr Surg 1982; 69: 110-116.
43. Silverman MK et al.: Recurrence rates of treated basal cell carcinomas. Part 2: curettage-electrodesiccation. J Dermatol Surg Oncol 1991; 17: 720-6.
44. Marcil I, Stern RS: Risk of developing a subsequent nonmelanoma skin cancer in patients with a history of nonmelanoma skin cancer: a critical review of the literature and meta-analysis. Arch Dermatol 2000; 136: 1524-30.
45. van Iersel CA et al.: Prognostic factors for a subsequent basal cell carcinoma: implications for follow up. Br J Dermatol 2005; 153:1078-80.
46. Park AJ, Strick M, Watson JD: Basal cell carcinomas: do they need to be followed up? J R Coll Surg Edinb 1994; 39: 109-11.
47. Smith SP, Grande DJ: Basal cell carcinoma recurring after radiotherapy: A unique difficult treatment subclass of recurrent basal cell carcinoma. J Dermatol Surg Oncol 1991; 17: 26.
48. Rowe DE, Carroll RJ, Day CL: Mohs surgery is the treatment of choice for recurrent (previously treated) basal cell carcinoma. J Dermatol Surg Oncol 1989; 15: 424-31.
49. Boulinguez S et al.: Histological evolution of recurrent basal cell carcinoma and therapeutic implications for incompletely excised lesions. Br J Dermatol 2004; 151: 623-6.
50. Mohs FE: Chemosurgery: A microscopically controlled method of cancer excision. Arch Surg 1941; 42: 279-295.
51. Nelson BR, Railan D, Cohen Scott Moh´s micrographic surgery for nonmelanoma skin cancers. Clin in Plast Surg 1997; 24(4): 705-718.
52. Rowe DE et al.: Long-term recurrence rates in previously untreated (primary) basal cell carcinoma: implications for patient follow-up. J Dermatol Surg Oncol 1989; 15: 315-28.
53. Malhotra R et al.: The Australian Mohs database, part II: periocular basal cell carcinoma outcome at 5-year follow-up. Ophthalmology 2004; 111: 631-6.
54. Leibovitch I et al.: Basal cell carcinoma treated with Mohs surgery in Australia II. Outcome at 5-year follow-up. J Am Acad Dermatol 2005; 53: 452-7.
55. Gaston DA, Naugle C, Clark DP: Mohs micrographic surgery referral patterns: the University of Missouri experience. Dermatol Surg 1999; 25: 862-6.
56. Cook J, Zitelli JA:Mohs micrographic surgery: a cost analysis. J Am Acad Dermatol 1998; 39: 698-703.
57. Hsuan JD, Harrad RA, Potts MJ, Collins C. Small margin excision of periocular basal cell carcinoma: 5 year results. Br J Ophthalmol 2004; 88: 358-60.
58. Smeets NWJ et al.: Surgical excision vsMohs´ micrographic surgery for basal-cell carcinoma of the face: randomised controlled trial. Lancet 2004; 364: 1766-72.
59. Conway RM, Themel S, Holbach LM: Surgery for primary basal cell carcinoma including the eyelid margins with intraoperative frozen section control: comperative interventional study with a minimum clinical follow up of 5 years. Br J Ophtalmol 2004; 88: 236-8.
60. Beirne GA, Beirne CG: Observations on the critical margin for the complete excision of carcinoma of the skin. Arch Dermatol 1959; 80: 344-5.
61. Bart RS et al.: Scalpel excision of basal cell carcinomas. Arch Dermatol 1978(114); V, 739-742.
62. Emmett AJJ, Broadbent GG: Basal cell carcinoma in Queensland. Aust NZ J Surg 1981; 51: 576-90.
63. Epstein E: How accurate is the visual assessment of basal carcinoma margins? Br J Dermatol 1973; 89: 37-43.
64. Bisson MAet al.: Do plastic surgeons resect basal cell carcinomas too widely? A prospective study comparing surgical and histological margins. Br J Plast Surg 2002; 55: 293-7.
65. Griffith BH, McKinney P: An appraisal of the treatment of basal cell carcinoma of the skin. Plast Rec Surg 1973; 51: 563-571.
66. Breuninger H et al.: The margin of safety and depth of excision in surgical treatment of a basalioma. Use of 3-dimensional histologic study of 2016 tumors. Hautarzt 1989; 40: 693-700.
67. Wolf DJ, Zitelli JA: Surgical margins for basal cell carcinoma. Arch Dermatol 1987; 123: 340-4 .
68. Griffiths RW, Suvarna SK, Stone J: Basal cell carcinoma histological clearance margins: an analysis of 1539 conventionally excised tumours. Wider still and deeper? J of Plast Rec and Aest Surgery 2007; 60: 41-47.
69. Collin JRO: Basal cell carcinoma in the eyelid region. Br J Ophtalmol 1976; 60: 806-809.
otrzymano/received: 2009-05-21 zaakceptowano/accepted: 2009-08-12 Adres/address: *Józef Jethon Department of Plastic Surgery, The Medical Centre of Postgraduate Education in Warsaw Czerniakowska 231 Str., 00-416 Warsaw tel.: (0-22) 584-11-91 e-mail: jjethon@szpital-orlowskiego.pl Pełna wersja artykułu Basal cell carcinoma. Current views (Part III). Surgical treatment dostępna w Czytelni Medycznej Borgis. |
Chcesz być na bieżąco? Polub nas na Facebooku: strona Wydawnictwa na Facebooku |








